
Market intelligence, announcements, interviews, and industry insights
Expert analysis across the global graphite ecosystem - from mine to anode.
+++++++++++++++++++++++
https://businessanalytiq.com/procurementanalytics/index/graphite-price-index/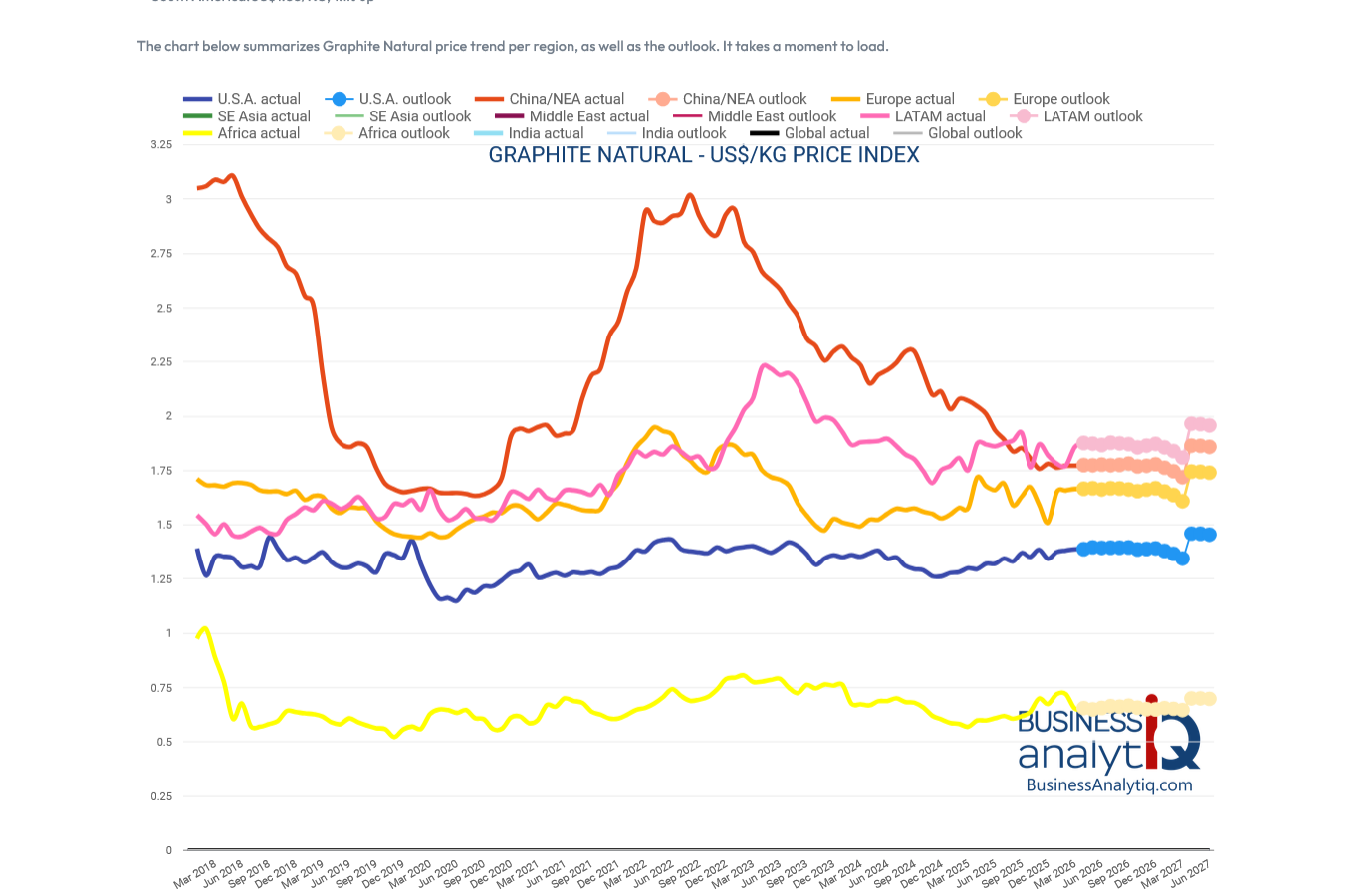
https://en.wikipedia.org/wiki/Graphite
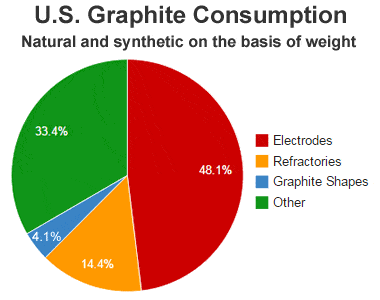
Graphite (/ˈɡræfaɪt/) is a crystalline allotrope (form) of the element carbon. It consists of many stacked layers of graphene, typically in excess of hundreds of layers. Graphite occurs naturally and is the most stable form of carbon under standard conditions. Synthetic and natural graphite are consumed on a large scale (1.3 million metric tons per year in 2022) for uses in many critical industries including refractories (50%), lithium-ion batteries (18%), foundries (10%), and lubricants (5%), among others (17%).[6] Graphite converts to diamond under extremely high pressure and temperature. Graphite's low cost, thermal and chemical inertness and characteristic conductivity of heat and electricity finds numerous applications in high energy and high temperature processes
https://www.eastcarb.com/uses-of-graphite/
For many years, graphite has been in existence and is used in various industries. In the modern world today, it has countless applications. In this article, we will discuss some of the most popular practical applications of graphite.


https://scienceinsights.org/what-is-graphite-used-for-in-industry-and-daily-life/
Graphite is used in everything from pencils and batteries to steelmaking and nuclear reactors. It’s one of the most versatile minerals on earth, valued for a unusual combination of properties: it conducts electricity and heat, resists extreme temperatures, and acts as a natural lubricant. That mix makes it essential across dozens of industries, some obvious and some you’d never guess.

https://geology.com/minerals/graphite.shtml
Graphite is a naturally occurring form of crystalline carbon. It is a native element mineral found in metamorphic and igneous rocks. Graphite is a mineral of extremes. It is extremely soft, cleaves with very light pressure, and has a very low specific gravity. In contrast, it is extremely resistant to heat and nearly inert in contact with almost any other material. These extreme properties give it a wide range of uses in metallurgy and manufacturing.

Flake graphite: Flake graphite produced in Madagascar.
Graphite is a mineral that forms when carbon is subjected to heat and pressure in Earth's crust and in the upper mantle. Pressures in the range of 75,000 pounds per square inch and temperatures in the range of 750 degrees Celsius are needed to produce graphite. These correspond to the granulite metamorphic facies.
+++++++++++
https://www.researchandmarkets.com/articles/key-companies-in-graphite
++++++++++++++++
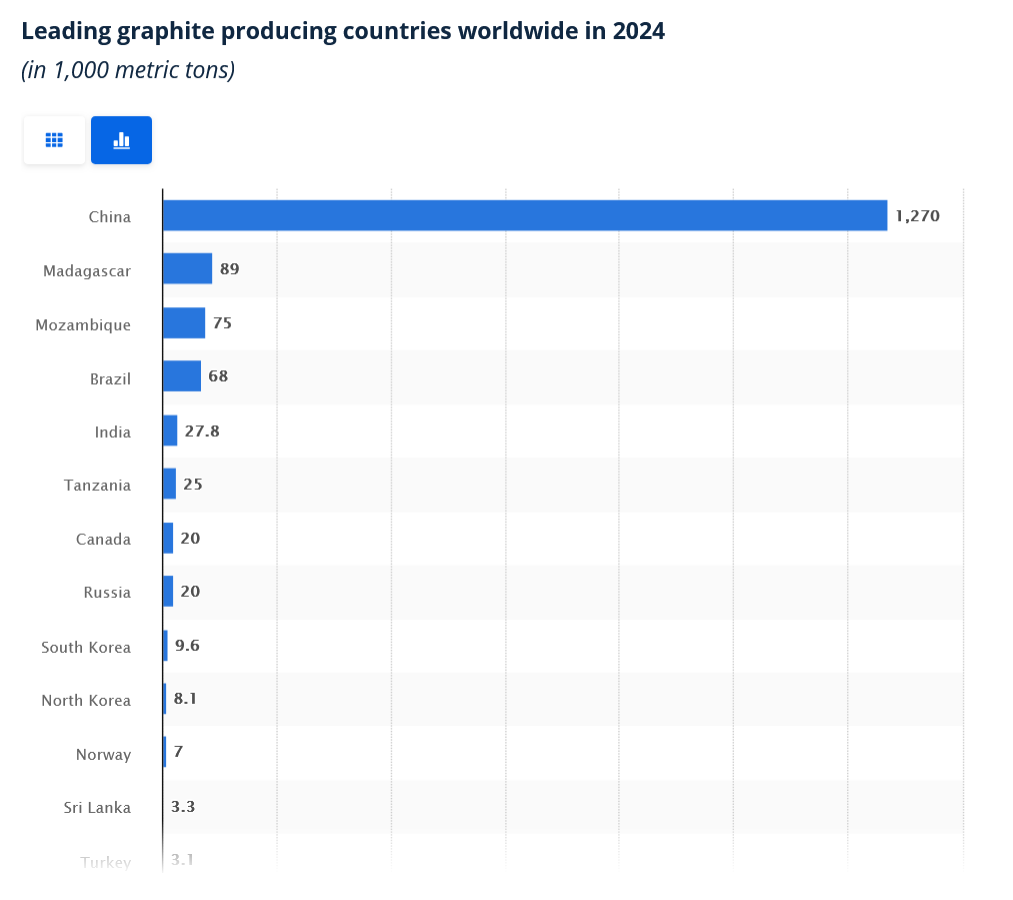
https://gotraysgraphite.com/top-ten-graphite-producing-countries/
+++++++++++++
Fullerene
https://en.wikipedia.org/wiki/Fullerene
A fullerene is an allotrope of carbon whose molecules consist of carbon atoms connected by single and double bonds so as to form a closed or partially closed mesh, with fused rings of five to six atoms. The molecules may have hollow sphere- and ellipsoid-like forms, tubes, or other shapes.
Fullerenes with a closed mesh topology are informally denoted by their empirical formula Cn, often written Cn, where n is the number of carbon atoms. However, for some values of n there may be more than one isomer.
The family is named after buckminsterfullerene (C60), the most famous member, which in turn is named after Buckminster Fuller. The closed fullerenes, especially C60, are also informally called buckyballs for their resemblance to the standard ball of association football. Nested closed fullerenes have been named bucky onions. Cylindrical fullerenes are also called carbon nanotubes or buckytubes.[1] The bulk solid form of pure or mixed fullerenes is called fullerite.[2]
Fullerenes had been predicted for some time, but only after their accidental synthesis in 1985 were they detected in nature[3][4] and outer space.[5][6] The discovery of fullerenes greatly expanded the number of known allotropes of carbon, which had previously been limited to graphite, diamond, and amorphous carbon such as soot and charcoal. They have been the subject of intense research, both for their chemistry and for their technological applications, especially in materials science, electronics, and nanotechnology.[7]
https://en.wikipedia.org/wiki/Carbon_nanotube
A carbon nanotube (CNT) is a tube made of carbon with a diameter in the nanometre range (nanoscale). They are one of the allotropes of carbon. Two broad classes of carbon nanotubes are recognized:
Carbon nanotubes can exhibit remarkable properties, such as exceptional tensile strength and thermal conductivity because of their nanostructure and strength of the bonds between carbon atoms. Some SWCNT structures exhibit high electrical conductivity while others are semiconductors. In addition, carbon nanotubes can be chemically modified. These properties are expected to be valuable in many areas of technology, such as electronics, optics, composite materials (replacing or complementing carbon fibres), nanotechnology (including nanomedicine), and other applications of materials science.
The predicted properties for SWCNTs were tantalising, but a path to synthesising them was lacking until 1993, when Iijima and Ichihashi at NEC, and Bethune and colleagues at IBM independently discovered that co-vaporising carbon and transition metals such as iron and cobalt could specifically catalyse SWCNT formation. These discoveries triggered research that succeeded in greatly increasing the efficiency of the catalytic production technique, and led to an explosion of work to characterise and find applications for SWCNTs.
216 pages and everything that you ever wanted to know about this topic!!

https://cdn.strategicmetalsinvest.com/wp-content/uploads/2025/02/mcs2025.pdf
Previous related threads:
Lithium Batteries, Cobalt and the Congo
Started by joj - Jan. 5, 2023, 7:53 p.m
. https://www.marketforum.com/forum/topic/91944/
++++++++++++++++++++++++++++
Rechargeable batteries
Started by metmike - July 11, 2023, 4:43 p.m.
https://www.marketforum.com/forum/topic/97099/
+++++++++++++++++
Energy transition is a hoax
Started by metmike - April 15, 2023, 5:50 p.m.
https://www.marketforum.com/forum/topic/94557/
++++++++++++++++++=
Lithium
Started by metmike - Sept. 16, 2025, 11:55 a.m.
https://www.marketforum.com/forum/topic/114700/#114703
https://www.visualcapitalist.com/visualizing-global-rare-earth-metals-production-1995-2023/

Sort of related:
Wind Farms
Started by wglassfo - May 11, 2022, 11:37 a.m.
https://www.marketforum.com/forum/topic/84297/
++++++++++++++
Killing Coal
Started by metmike - Nov. 21, 2021, 10:57 p.m.
https://www.marketforum.com/forum/topic/78168/
+++++++++++++++++++
Wind/ solar/batteries
By metmike - April 19, 2026, 7:31 p.m.
Life without petroleum based products: 6,000 products made with petroleum. Killing Coal. Fossil fuels and fertilizer. Biden praises high gasoline prices.
https://www.marketforum.com/forum/topic/84689/

Just in case you think we could go to Net-Zero oil any time soon:
Crude oil is legally treated as a mineral, and that classification shapes who owns it, how rights are transferred, and how it's taxed.
https://legalclarity.org/is-crude-oil-a-mineral-what-the-law-says/
Crude oil fails the geological definition of a mineral, but for legal and tax purposes, it is classified as one. Federal law explicitly lists oil alongside coal, phosphate, and other mineral deposits as resources subject to government leasing and regulation.1 That legal classification drives real-world consequences: it determines who owns underground oil, who can extract it, how royalty income is taxed, and what happens when surface owners and mineral owners disagree.
Geologists define a mineral using five requirements. The substance must be naturally occurring, inorganic, solid, have a defined chemical composition, and possess an orderly crystalline structure. Crude oil fails on at least three counts. It is a liquid, not a solid. It formed from the remains of ancient marine organisms like plankton and algae, making it organic rather than inorganic. And it has no crystal structure at all.